|
11/4/2023 0 Comments Definition of sigma bondA tertiary carbon has three carbon neighbors.A secondary carbon has two carbon neighbors.A primary carbon has one carbon neighbor.Carbon atoms in a molecule are categorized by the number of carbon neighbors they have: This coupled with the strength of the carbon–carbon bond gives rise to an enormous number of molecular forms, many of which are important structural elements of life, so carbon compounds have their own field of study: organic chemistry.īranching is also common in C−C skeletons. Chains and branching Ĭarbon is one of the few elements that can form long chains of its own atoms, a property called catenation. The use of the p-orbitals forms a pi bond. A triple bond is formed with an sp-hybridized orbital and two p-orbitals from each atom. A double bond is formed with an sp 2-hybridized orbital and a p-orbital that is not involved in the hybridization. Carbon atoms can also form double bonds in compounds called alkenes or triple bonds in compounds called alkynes. 
In fact, the carbon atoms in the single bond need not be of the same hybridization. In ethane, the orbitals are sp 3- hybridized orbitals, but single bonds formed between carbon atoms with other hybridizations do occur (e.g. 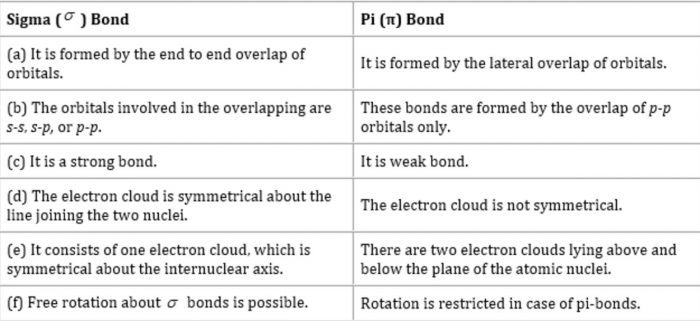
The carbon–carbon single bond is a sigma bond and is formed between one hybridized orbital from each of the carbon atoms. The most common form is the single bond: a bond composed of two electrons, one from each of the two atoms. 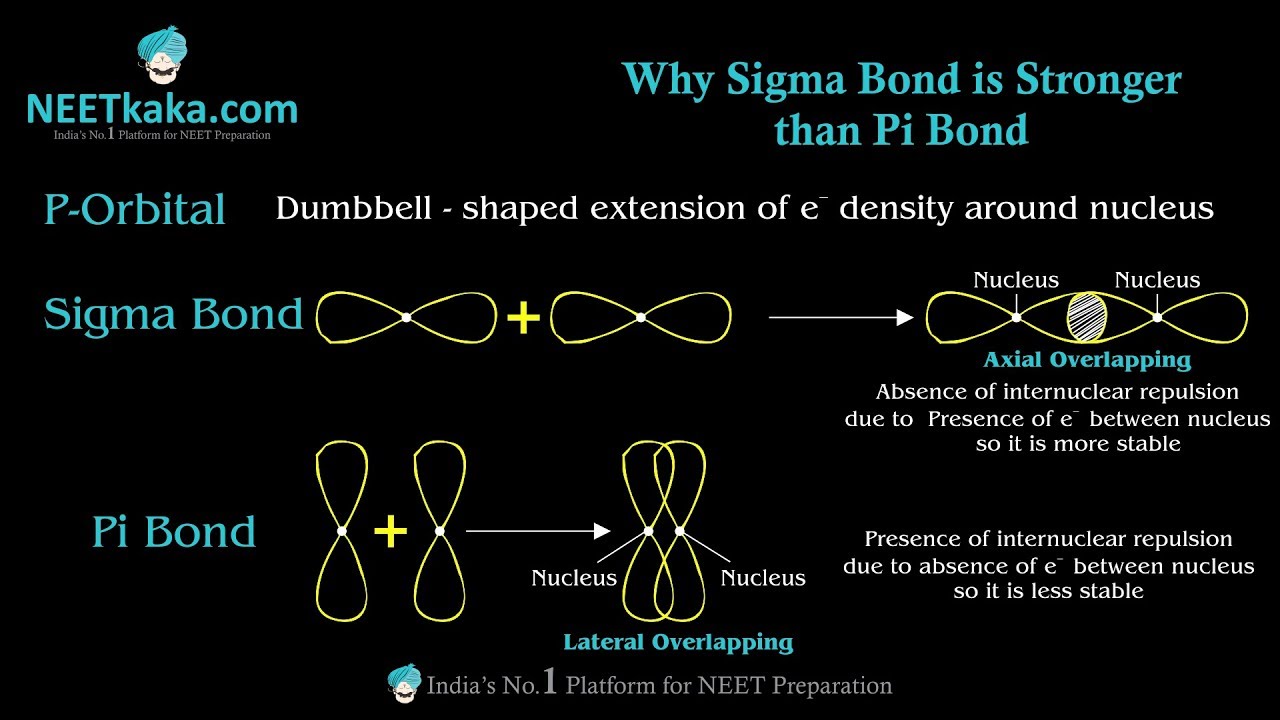
A carbon–carbon bond is a covalent bond between two carbon atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
